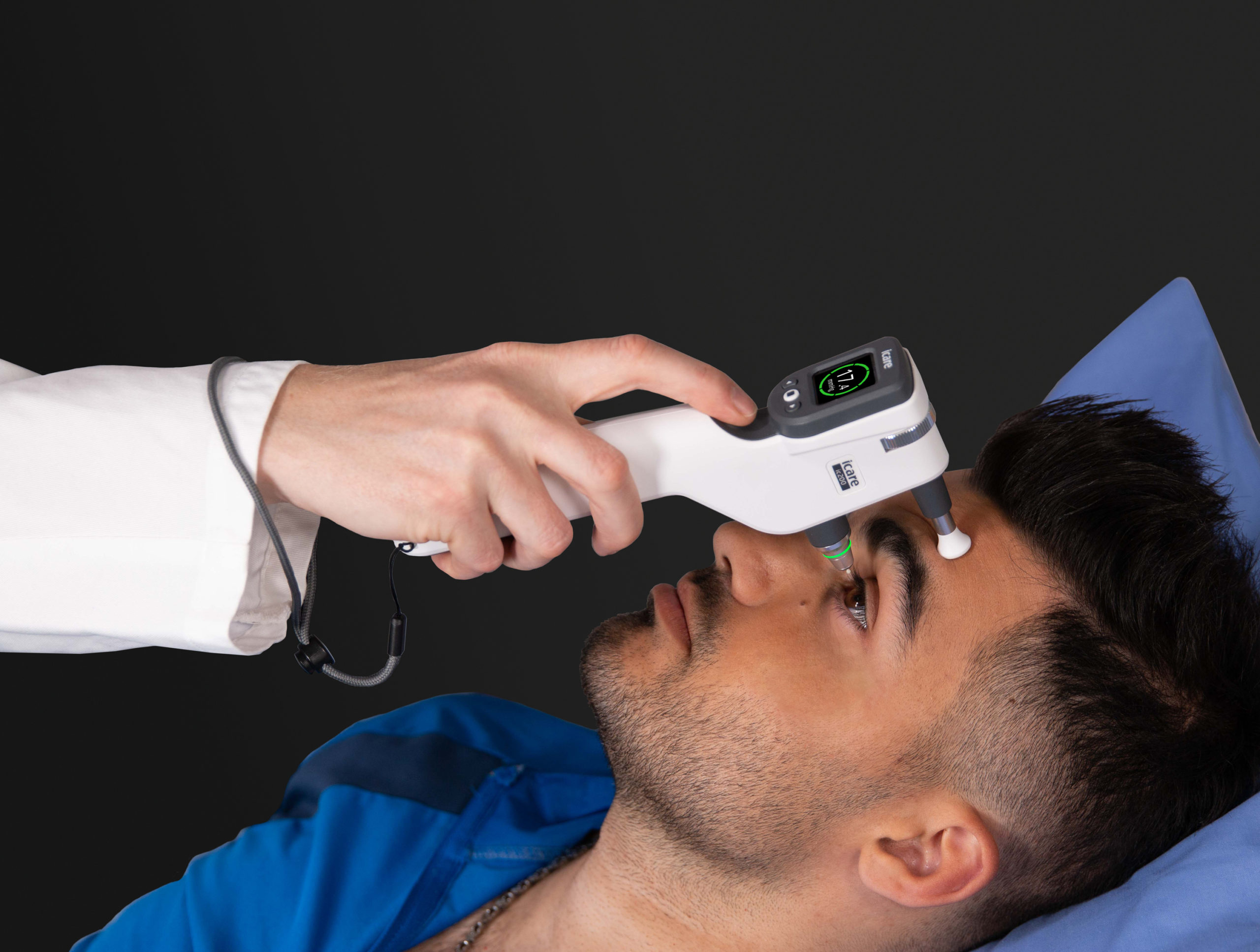
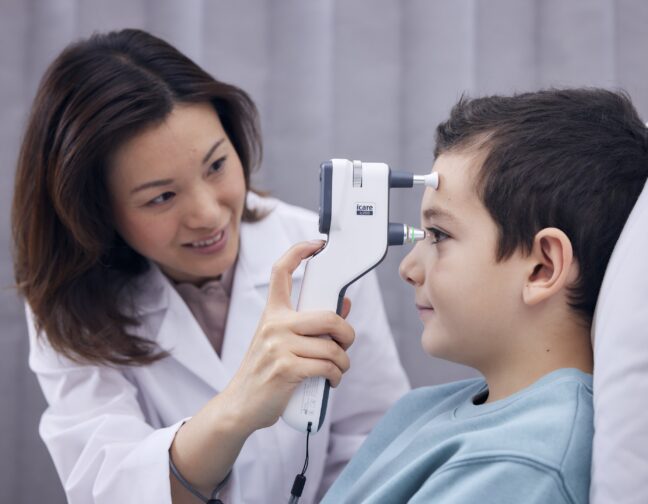
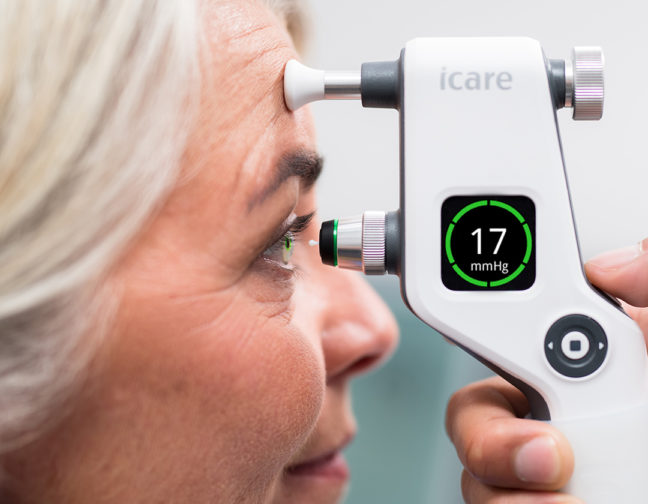
The safe, easy-to-use, hand-held iCare tonometers enable the measurement of IOP in a process that is free from air puffs, microaerosol formation, anesthetic drops, or any risk of cross contamination.
With the world grappling with the coronavirus pandemic, it is crucial to maintain strict hygiene and social distancing standards in all medical settings to prioritize the safety of both healthcare workers and patients. iCare’s patented rebound technology which uses disposable probes is a proven safe and hygienic way of obtaining accurate IOP measurements. This makes the iCare tonometers an ideal choice for eyecare professionals in current times.
How does the innovative technology and unique design of iCare rebound tonometers significantly reduce the risk of infectious transmissions compared to traditional tonometry solutions?
Single-use probes eliminate risk of cross contamination
While measuring intraocular pressure, many conventional non-contact tonometry solutions in the market lead to the generation of microaerosol particles, tear film spread, and the risk of cross contamination. The iCare rebound tonometers use individually packed, hygienic, single-use probes that protect against microbiological cross-contamination and infectious transmissions between patients.
As one of the only handheld tonometers in the market that use disposable probes, the iCare tonometers are a preferred choice to comply with the safety and hygiene standards that the epidemic situation demands. The iCare tonometers are also easy to clean before and after every use.
No air puffs — no microaerosol spreading!
Scientific studies* suggest that COVID-19 can spread through microaerosol transmission. Noncontact tonometers (NCT) that use air puff tonometry to measure the eye’s intraocular pressure cause the tear film of the eye to spread, increasing the risk of cross-contamination.
The iCare tonometer is being recognized as an advantageous way to measure IOP because the single-use probe allows eye pressure to be measured without leading to potentially dangerous microaerosol spreading.
No need for anesthesia — pain-free, safe, and quick
One of the biggest advantages of iCare tonometers is the ease of use. The smartly designed devices let you measure intraocular pressure without the need for anesthetic drops, or air.
The touch of the disposable probe is so quick that the measurement is barely noticed, even by children or otherwise non-compliant patients.
At home measurement for optimum safety and social distancing
The iCare HOME2 tonometer enables for the patients to measure their intraocular pressure at home during normal daily activities. The user-friendly technology is so easy to use that patients can perform the measurement themselves with professional accuracy.
During a pandemic, when eyecare clinics could be limiting appointments to reduce the risk of transmission, the iCare HOME2 is a perfect solution. It is designed for easy self-measuring and offers results as accurate as those of professional models, with the added advantage of providing ample, real-world IOP data. It offers the convenience of home measurements to patients who are unable to or anxious about attending clinic appointments.
Patients can easily and accurately monitor their IOP at different times of the day and share readings with their eye health practitioners for review, while following social distancing restrictions.
During these challenging epidemic times when there is a need to adhere to strict hygiene and social distancing standards, iCare tonometers offer an effective, accurate, and safe IOP measurement solution.
* References:
1. Britt JM, Clifton BC, Barnebey HS, Mills RP. Microaerosol formation in noncontact “air-puff” tonometry. Arch Ophthalmol. 1991;109:225–228.
2. Tracy H. T. Lai, Emily W. H. Tang, Sandy K. Y. Chau, Kitty S. C. Fung, Kenneth K. W. Li. Stepping up infection control measures in ophthalmology during the novel coronavirus outbreak: an experience from Hong Kong. Graefe’s Archive for Clinical and Experimental Ophthalmology. 2020; e-published 03 March 2020.